An in depth look at the role of milk proteins on the foaming process

In the foaming process, caseins and whey proteins play a fundamental role since they absorb at the air-liquid interfaces forming a film around the air bubbles stabilising them.
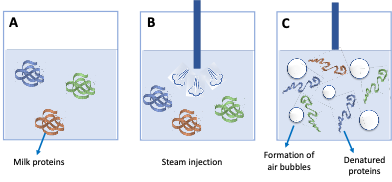
The role of milk proteins on the foaming process and bubble stabilisation can be summarised as follows: before steam injection, milk proteins are present in the milk in their native (folded) conformation in which they are stable and active (Figure 1 A).
As soon as the gas is introduced into the milk (Figure 1B), a gas-liquid interfacial film is formed stabilised by the surface tension between the gas and the liquid (milk) promoting the foam formation (Figure 1C).
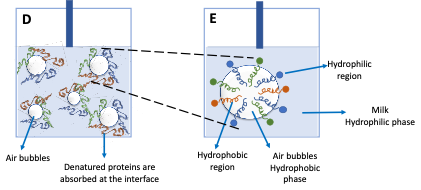
Denatured (unfolded) proteins orientate their hydrophilic and hydrophobic groups at the air-liquid interface (Figure 1D), where the polar groups are rearranged towards the aqueous phase and the non-polar group towards the non-aqueous (Figure 1E). The subsequent interactions among unfolded proteins give rise to a strong, viscous, and elastic interfacial film that stabilises the air bubbles.
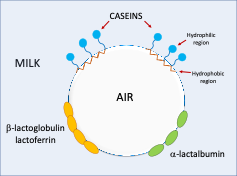
In order to obtain a quality cappuccino foam, it is important to know and study the behaviour of milk proteins involved in the formation and stabilisation of the foam itself. Caseins play an important role with milk proteins and their ability to form a stable film on the air-liquid interface because they are less structured and more flexible than the globular whey proteins. This means they can occupy a larger area at the interface which results in a soft and compressible texture.
β-casein and αs1-casein are present in the milk foam and their concentration does not vary when the milk is treated by the coffee machine steamer in the temperature range of 50-80°C. It is known that β-casein diffuses more extensively at the air-liquid interface thanks to the hydrophobic amino acid region anchored at the layer surface and the hydrophilic region directed towards the aqueous phase.
αs1-casein has a smaller distribution at the air-liquid interface and forms more voluminous and stable foam with respect to β-casein but is not able to form a strong interfacial layer. Unlike caseins, whey proteins have a globular structure, including β-lactoglobulin, α-lactalbumin, bovine serum albumin, lactoferrin, immunoglobulins, and other minor proteins. The difference between the soft, unstructured caseins and more structured whey proteins is responsible for the foaming properties of milk.
β-lactoglobulin is known to exist in two main genetic variants namely A and B, both responsible for the foam stability thanks to their ability to form intermolecular disulphide bonds at the interface. In our work we found that β-lactoglobulin variants A and B showed a decreased concentration in the milk foam in the temperature range of 50-80°C after steam injection treatment, due to the formation of unfolded protein that allows hydrophobic interactions and the compaction of the protein molecules at the interface. This results in a strong interfacial film and good bubble coverage.
Similarly, we found that lactoferrin concentration decreased in the milk foam after steam injection treatment because of the possible formation of denatured protein aggregates or the formation of complexes with one or more compounds derived from the coffee. From literature, it is known that lactoferrin can form complexes with some polyphenols such as chlorogenic acid leading to better foamability and foam stability than the lactoferrin alone.
The concentration of milk foam α-lactalbumin is not affected by the increasing temperature after the steam injection treatment and probably could not compact well at the interface to ensure the necessary film and foams stabilisation. It has been proved, in fact, that α-lactalbumin, is more structured than β-lactoglobulin and can form a voluminous but unstable foam.
The model shown below summarises the role of the milk proteins in the foam formation and stability.
Become part of a global community of coffee professionals. Access courses and be the first to hear about news, research and events from across the coffee world.
Join now
© 2024 Coffee Knowledge Hub
Simonelli Group SpA
Via Emilio Betti, 1, 62020
Belforte del Chienti MC
P.IVA 01951160439
VAT n. 01951160439
info@coffeeknowledgehub.com

This website utilises cookies to enable necessary site functionality such as logging you in to your account. By remaining on this website you indicate your consent as outlined in our Cookie Policy.